- Home
- About
- Contact
- Final cut pro 10-4-2 downloads
- Act of war direct action high treason
- Kailasanathan actress parvathi
- Change domain functional level fortinet vpn issues
- Beersmith 2 vs 3
- Album tamil movie mp3 songs free download
- Free microsoft excel add ins
- Addic7ed empire season 2 episode 1
- Rome total war 2 battle
- Prison break season 1 download free
- Tyt uv8000e speaker mic
- Battlebots bracket
- Corner case for fpga simulation
- Nito installer apple tv 2 windows how long does it take
- Pbp3 form
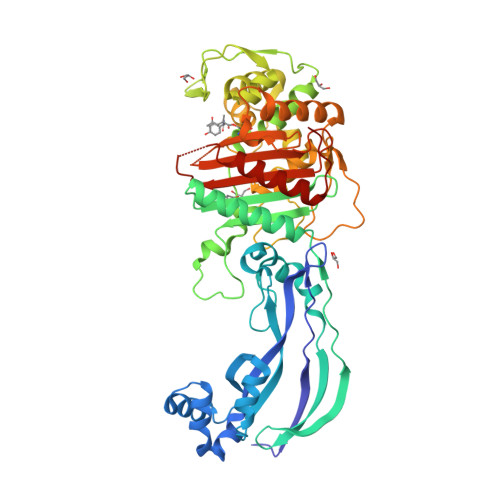
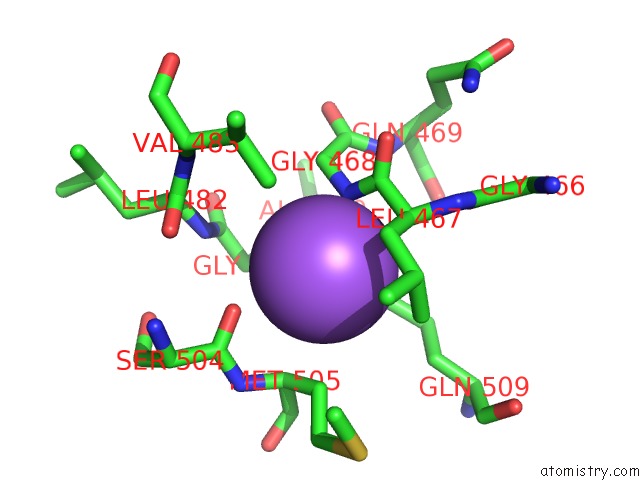
aeruginosa and related Gram-negative bacteria are sought and our study provides templates to assist that process and allows us to discuss new ways of inhibiting PBPs. DD-transpeptidase, also known as the cell division protein ftsI, is an essential protein that localizes at the septum where it forms a ternary complex with other cell division proteins.(Dasgupta. Rakita andHenryRosen Departments ofMedicine, University ofWashington, Seattle, Washington 98195, andSwedish HospitalMedicalCenter, Seattle, Washington 98104 Abstract Myeloperoxidase (MPO),H202,andchloride compriseapo- tent antimicrobial systembelieved to contribute to theantimi. Improved drugs to combat infections by P. Obtaining PBP3 in the form of a catalytically active water-soluble derivative suitable for X-ray analysis is an important aim of the research (Bartholom-De. Penicillin-binding Protein Inactivation byHumanNeutrophil Myeloperoxidase RobertM. Fraipont C, Alexeeva S, Wolf B, van der Ploeg R, Schloesser M, den Blaauwen T, Nguyen-Disteche M: The integral membrane FtsW protein and peptidoglycan synthase PBP3 form a subcomplex in Escherichia coli. (DSHB Hybridoma Product PBP3) Storage and Handling Recommendations Although many cell products are maintained at 4☌ for years without loss of activity, shelf-life at 4☌ is highly variable. High-resolution structures of the apo and ceftazidime-bound forms were obtained as well as complexes with meropenem, imipenem, and aztreonam. aeruginosa PBP3 (PaPBP3) and its -lactam recognition elements, we solved multiple X-ray structures of a soluble form of PaPBP3. aeruginosa is an important human pathogen, the structural data reveal the mode of action of the frontline antibiotic ceftazidime at the molecular level. PBP3 was deposited to the DSHB by Nardi, J. To help elucidate the molecular mechanism of P. The conserved binding mode of β-lactam-based inhibitors appears to extend to other PBPs, as suggested by a comparison of the PBP3/ceftazidime complex and the Escherichia coli PBP1b/ceftoxamine complex. The orientations of the two β-lactams in the active site and the key interactions formed between the ligands and PBP3 are similar despite differences in the two drugs, indicating a degree of flexibility in the binding site. PBP3 interacts with PBP1B and both form, and are likely functional as, homodimers (Zijderveld et al., 1991 Bertsche et al., 2005, 2006 Sauvage et al., 2014). Binding of either carbenicillin or ceftazidime to purified PBP3 increases the thermostability of the enzyme significantly and is associated with local conformational changes, which lead to a narrowing of the substrate-binding cleft. Overall, the structures of apo and acyl complexes are very similar however, variations in the orientation of the amino-terminal membrane-proximal domain relative to that of the carboxy-terminal transpeptidase domain indicate interdomain flexibility. We report the first crystal structures of a penicillin-binding protein (PBP), PBP3, from Pseudomonas aeruginosa in native form and covalently linked to two important β-lactam antibiotics, carbenicillin and ceftazidime.
